
Europe and ROW
Full Safety Information
Translated key words:
| Atresia | The absence, or the closure, of an opening, passage, or cavity. |
| Barrett’s esophagus | A condition in which the cells lining the lower part of the esophagus have changed or been replaced with abnormal cells that could lead to cancer of the esophagus. |
| Cardiopulmonary | Heart and lungs |
| Cirrhosis | A chronic disease of the liver characterized by the replacement of normal tissue with scar tissue and the loss of functional liver cells. |
| Cholecystectomy | Removal of the gallbladder. |
| Contraindicated | A treatment, procedure, or activity should not be done or used in this situation for medical reasons. |
| Crohn’s disease | A chronic inflammatory disease, primarily involving the small and large intestine, but which can affect other parts of the digestive system as well. It can lead to abdominal pain, severe diarrhea, fatigue, weight loss and malnutrition. |
| Dysphagia | Difficulty or discomfort in swallowing. |
| Distension | Stretched out |
| Duodenal ulceration | Irritation/injury of the inside wall of the small intestine |
| Dysmotility | Diseases of the muscles of the gastrointestinal tract (esophagus, stomach, small and large intestines) in which the muscles do not work normally. |
| Esophageal Varices | Swollen veins in the lower esophagus. |
| Esophagitis | Inflammation of the upper part of the intestine connecting mouth to stomach |
| Explantation | Taking out of the body |
| Fundoplication | Surgical operation of wrapping part of the fundus of the stomach around the base of the esophagus |
| Gastric | Stomach |
| Gastric ulceration | Irritation/injury of the inside wall of the stomach |
| Gastric varices | Swollen veins in the stomach |
| Gastrointestinal tract | Both stomach and intestine |
| Hernia | A general term used to describe a bulge or protrusion of an organ through the structure or muscle that usually contains it. |
| hypertension | Abnormally high blood pressure |
| Intestine | A long, continuous tube running from the stomach to the anus. Most absorption of nutrients and water happen in the intestines. The intestines include the small intestine, large intestine, and rectum |
| Laparotomy | Surgical incision of the abdominal wall |
| Lupus erythematosus | Skin inflammation |
| Pancreatitis | Inflammation of the pancreas |
| Percutaneous | Through the skin |
| Polyneuropathies | A condition in which a person’s nerves that run throughout your body are damaged. It affects the nerves in your skin, muscles, and organs. When nerves are damaged, they can’t send regular signals back to your brain. |
| Reflux | Flow back or return. Gastroesophageal reflux is when what’s in your stomach backs up into your esophagus. |
| Revision procedure | Revision weight loss surgery is a surgical procedure that is performed on patients who have already undergone a form of bariatric surgery, and have either had complications from such surgery or have not achieved significant weight loss results from the initial surgery. |
| Splenectomy | Removal of the spleen |
| Stenoses | Narrowing of the diameter of the GI track |
| Sutured | Sewn in with at thread |
| Telangiectases | Spider veins |
| Trocar | A surgical instrument with a three-sided cutting point enclosed in a tube. Trocars are placed through the abdomen during laparoscopic surgery. |
INTENDED USE / INDICATIONS
The Lap-Band® System is indicated for use in weight reduction for severely obese patients with a Body Mass Index (BMI) of at least 35 or a BMI of at least 30 with one or more severe comorbid conditions, or those who are 100 pounds or more over their estimated ideal weight according to the 1983 Metropolitan Life Insurance Tables (use the midpoint for medium frame). It is indicated for use only in severely obese patients 14 years and older who have failed more conservative weight-reduction alternatives such as supervised diet, exercise and behavior modification programs. Patients who elect to have this surgery must make the commitment to accept significant changes in their eating habits for the rest of their lives.
Weight loss associated with the Lap-Band® System has been shown to improve or lead to remission of type 2 diabetes in patients with BMI greater than or equal to 35.
CONTRAINDICATIONS
The Lap-Band AP® System is contraindicated in:
- Patients with inflammatory diseases of the gastrointestinal tract (part of the digestive system consisting of the stomach and intestines), including severe intractable esophagitis, gastric ulceration, duodenal ulceration, or specific inflammation such as Crohn’s disease.
- Patients with severe cardiopulmonary diseases or other serious organic disease which may make them poor surgical candidates.
- Patients with potential upper gastrointestinal bleeding conditions such as esophageal or gastric varices or congenital or acquired intestinal telangiectases.
- Patients with portal hypertension.
- Patients with congenital or acquired anomalies of the GI tract such as atresias or stenoses.
- Patients who have/experience an intra-operative gastric injury during the implantation procedure, such as a gastric perforation at or near the location of the intended band placement.
- Patients with cirrhosis.
- Patients with chronic pancreatitis.
- Patients who are addicted to alcohol and/or drugs.
- Patients under 14 years of age (adolescent patients 14 to 17 years of age must be post-pubescent).
- Patients who have an infection anywhere in their body or where the possibility of contamination prior to or during the surgery exists.
- Patients on chronic, long-term steroid treatment.
- Patients who are unable or unwilling to comply with dietary restrictions that are required by this procedure.
- Patients who are known to have, or suspected to have, an allergic reaction to materials contained in the system or who have exhibited pain intolerance to implanted devices.
- Patients or family members with a known diagnosis or pre-existing symptoms of autoimmune connective-tissue disease such as systemic lupus erythematosus or scleroderma.
- Pregnancy: Placement of the Lap-Band AP® System is contraindicated for patients who currently are or may be pregnant. Patients who become pregnant after band placement may require deflation of their bands.
WARNINGS
- Laparoscopic or laparotomic placement of the Lap-Band AP® System is major surgery and death can occur.
- Failure to secure the band properly may result in its subsequent displacement and necessitate a second operation.
- A large hiatal hernia may prevent accurate positioning of the device. Placement of the band should be considered on a case-by-case basis depending on the severity of the hernia.
- The band should not be sutured to the stomach. Suturing the band directly to the stomach may result in erosion.
- Patients’ emotional and psychological stability should be evaluated prior to surgery. Gastric banding may be determined by a physician to be inappropriate for select patients.
- Patients should be advised that the Lap-Band AP® System is a long-term implant. Explant (removal) and replacement surgery may be indicated at any time. Medical management of adverse reactions may include explantation. Revision surgery for explantation and replacement may also be indicated to achieve patient satisfaction.
- Esophageal distension or dilatation has been reported to result from stoma obstruction from over-restriction by excessive band inflation. Patients should not expect to lose weight as fast as gastric bypass patients, and band inflation should proceed in small increments. Deflation of the band is recommended if esophageal dilatation develops.
- Some types of esophageal dysmotility may result in inadequate weight loss or may result in esophageal dilatation when the band is inflated and may require removal of the band. On the basis of each patient’s medical history and symptoms, surgeons should determine whether esophageal motility function studies are necessary. If these studies indicate that the patient has esophageal dysmotility, the increased risks associated with band placement must be considered.
- Patients with Barrett’s esophagus may have problems associated with their esophageal pathology that could compromise their post-surgical course. Use of the band in these patients should be considered on the basis of each patient’s medical history and severity of symptoms.
- Patient self-adjustment of superficially placed access ports has been reported. This can result in inappropriate band tightness, infection and other complications.
- Patients must be carefully counseled on the need to report all vomiting, abdominal pain or other gastrointestinal or nutritional issues as these symptoms may indicate a condition not related to the Lap-Band® System.
PRECAUTIONS
- Laparoscopic band placement is an advanced laparoscopic procedure. Surgeons planning laparoscopic placement must:
- Have extensive advanced laparoscopic experience, i.e., fundoplications.
- Have previous experience in treating obese patients and have the staff and commitment to comply with the long-term follow-up requirements of obesity procedures.
- Participate in a training program for the Lap-Band® System authorized by ReShape Lifesciences, Inc. or an authorized ReShape Lifesciences, Inc. distributor (this is a requirement for use).
- Be observed by qualified personnel during their first band placements.
- Have the equipment and experience necessary to complete the procedure via laparotomy if required.
- Be willing to report the results of their experience to further improve the surgical treatment of severe obesity.
- It is the responsibility of the surgeon to advise the patient of the known risks and complications associated with the surgical procedure and implant.
- As with other gastroplasty surgeries, particular care must be taken during dissection and during implantation of the device to avoid damage to the gastrointestinal tract. Any damage to the stomach during the procedure may result in erosion of the device into the GI tract.
- During insertion of the Calibration Tube, care must be taken to prevent perforation of the esophagus or stomach.
- Revision procedures may require the existing staple line to be partially disrupted to avoid having a second point of obstruction below the band. As with any revision procedure, the possibility of complications such as erosion and infection is increased. Any damage to the stomach during the procedure may result in peritonitis and death or in late erosion of the device into the GI tract.
- Care must be taken to place the Access Port in a stable position away from areas that may be affected by significant weight loss, physical activity or subsequent surgery. Failure to do so may result in the inability to perform percutaneous band adjustments.
- Care must be taken during band adjustment to avoid puncturing the tubing that connects the Access Port and band, as this will cause leakage and deflation of the inflatable section.
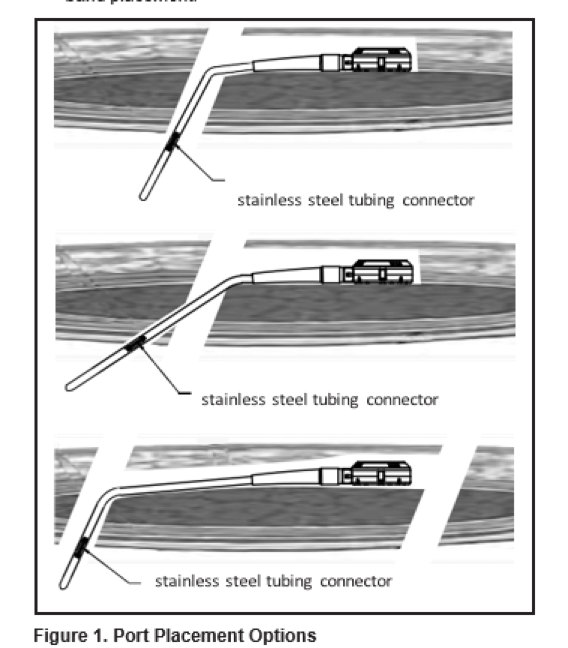
- Failure to create a stable, smooth path for the Access Port tubing, without sharp turns or bends, can result in tubing breaks and leakage. In order to avoid incorrect placement, the port should be placed lateral to the trocar opening. A pocket must be created for the port so that it is placed far enough from the trocar path to avoid abrupt kinking of the tubing. The tubing path should point in the direction of the Access Port connector so that the tubing will form a straight line with a gentle arching transition into the abdomen. (See Figure 1. Port Placement Options).
- The Lap-Band AP® System is for single use only. Do not use a band, Access Port, needle or calibration tube that appears damaged (cut, torn, etc.) in any way. Do not use any of the above components if the package has been opened or damaged or if there is any evidence of tampering. If packaging has been damaged, the product may not be sterile and may cause an infection.
- Do not attempt to clean or re-sterilize any part of the Lap-Band AP® System. The product may be damaged or distorted if re-sterilized.
- Special care must be used when handling the device because contaminants such as lint, fingerprints and talc may lead to a foreign body reaction.
- Care must be taken to avoid damaging the band, its inflatable section or tubing, the Access Port or the calibration tube. Use only rubber-shod clamps to clamp tubing.
- The band, Access Port and calibration tube may be damaged by sharp objects and manipulation with instruments. A damaged device must not be implanted. For this reason, a stand-by device should be available at the time of surgery.
- Failure to use the tubing end plug during placement of the band may result in damage to the band tubing during band placement.
- Do not push the tip of any instrument against the stomach wall or use excessive electrocautery. Stomach perforation or damage may result in peritonitis and death.
- Over-dissection of the stomach during placement may result in slippage or erosion of the band and require reoperation.
- Failure to use an appropriate atraumatic instrument to lock the band may result in damage to the band or injury to surrounding tissues.
- When adjusting band volume, take care to ensure the radiographic screen is perpendicular to the needle shaft (the needle will appear as a dot on the screen). This will facilitate adjustment of needle position as needed while moving through the tissue to the port.
- When adjusting band volume, use of an inappropriate needle may cause Access Port leakage and require reoperation to replace the port. Use only Lap-Band AP® System Access Port Needles. Do not use standard hypodermic needles, as these may cause leaks.
- When adjusting band volume, the needle must be inserted perpendicular to the Access Port septum. Failure to do so may cause damage to the port and result in leaks.
- When adjusting band volume never enter the Access Port with a “syringeless” needle. The fluid in the device is under pressure and will be released through the needle.
- When adjusting band volume after the septum is punctured, do not tilt or rock the needle, as this may cause fluid leakage or damage to the septum.
- When adjusting band volume, if fluid has been added, it is important to establish that the stoma is not too small before discharge. Care must be taken to not add too much saline, thereby closing the gastric stoma. Check the adjustment by having the patient drink water. If the patient is unable to swallow, remove some fluid from the port, then re-check. A physician familiar with the adjustment procedure must be available for several days post-adjustment to deflate the band in case of an obstruction.
- It is the responsibility of the surgeon to advise the patient of the dietary restrictions that follow this procedure and to provide diet and behavior modification support. Failure to adhere to the dietary restrictions may result in obstruction and/or failure to lose weight.
- Patients must be carefully counseled on the need for proper dietary habits. They should be evaluated for nutritional (including caloric) needs and advised on the proper diet selection. The physician may choose to prescribe appropriate dietary supplements. Appropriate physical monitoring and dietary counseling should take place regularly.
- Patients must be cautioned to chew their food thoroughly. Patients with dentures must be cautioned to be particularly careful to cut their food into small pieces. Failure to follow these precautions may result in vomiting, stomal irritation and edema, possibly even obstruction.
- Patients must be seen regularly during periods of rapid weight loss for signs of malnutrition, anemia or other related complications.
- Anti-inflammatory agents, such as aspirin, and non- steroidal anti-inflammatory drugs (NSAIDs), may irritate the stomach and should be used with caution. The use of such medications may be associated with an increased risk of erosion.
- Patients who become pregnant, severely ill, or who require more extensive nutrition may require deflation of their bands.
- All patients should have their reproductive areas shielded during radiography.
- Insufficient weight loss may be caused by pouch enlargement or, more infrequently, band erosion in which case further inflation of the band would not be appropriate.
- Elevated homocysteine levels have been found in patients actively losing weight after obesity surgery. Supplemental folate and vitamin B12 may be necessary to maintain normal homocysteine levels. Elevated homocysteine levels may increase cardiovascular risk and the risk of neural tube abnormalities.
- Although there have been no reports of autoimmune disease with the use of the Lap-Band® System, auto-immune diseases/connective tissue disorders (i.e., systemic lupus erythematosus, sclero-derma) have been reported following long-term implantation of other silicone implants. However, there is no conclusive evidence to substantiate a relationship between connective-tissue disorders and silicone implants.
ADVERSE EVENTS
It is important to discuss all possible complications and adverse events with your patient. Complications which may result from the use of this product include the risks associated with the medications and methods utilized in the surgical procedure, the risks associated with any surgical procedure and the patient’s degree of intolerance to any foreign object implanted in the body.
Perforation of the stomach can occur. Death can also occur. Specific complications of laparoscopic surgery can include spleen damage (sometimes requiring splenectomy) or liver damage, bleeding from major blood vessels, lung problems, thrombosis, and rupture of the wound.
Ulceration, gastritis, gastroesophageal reflux, heartburn, gas bloat, dysphagia, dehydration, constipation, and weight regain have been reported after gastric restriction procedures.
Band slippage and/or pouch dilatation can occur. Gastroesophageal reflux, nausea and/or vomiting with early or minor slippage may be successfully resolved by band deflation in some cases. More serious slippages may require surgery to reposition and/or remove the band. Immediate reoperation to remove the band is indicated if there is total stoma outlet obstruction that does not respond to band deflation or if there is abdominal pain.
Gastric banding done as a revision procedure has a greater risk of complications. Prior abdominal surgery is commonly associated with adhesions involving the stomach. In the
U.S. pivotal study of severely obese adults, 42% of the subjects undergoing revision surgery were reported to have adhesions involving the stomach. Care and time must be taken to adequately release the adhesions to provide access, exposure and mobilization of the stomach for a revision procedure.
There is a risk of band erosion into stomach tissue. Erosion of the band into stomach tissue has been associated with revision surgery after the use of gastric-irritating medications, after stomach damage and after extensive dissection or use of electrocautery, and during early surgeon experience. Symptoms of band erosion may include reduced weight loss, weight gain, Access Port infection, or abdominal pain. Reoperation to remove the device is required.
Reoperation for band erosions may result in a gastrectomy of the affected area. Eroded bands have been removed gastroscopically in very few cases. Consultation with other experienced Lap-Band® System surgeons is strongly advised in these cases.
Esophageal distension or dilatation has been infrequently reported. This is most likely a consequence of incorrect band placement, over-restriction or stoma obstruction. It can also be due to excessive vomiting or patient noncompliance, and may be more likely in cases of pre-existing esophageal dysmotility. Deflation of the band is recommended if esophageal dilatation develops. A revision procedure may be necessary to reposition or remove the band if deflation does not resolve the dilatation.
Obstruction of stomas has been reported as both an early and a late complication of this procedure. This can be caused by edema, food, improper initial calibration, band slippage, pouch torsion, or patient non-compliance regarding choice and chewing of food.
Infection can occur in the immediate post-operative period or years after insertion of the device. In the presence of infection or contamination, removal of the device is indicated.
Unplanned deflation of the band may occur due to leakage from the band, the port or the connecting tubing.
Nausea and vomiting may occur, particularly in the first few days after surgery and when the patient eats more than recommended. Nausea and vomiting may also be symptoms of stoma obstruction or a band/stomach slippage. Frequent, severe vomiting can result in pouch dilatation, stomach slippage or esophageal dilatation. Deflation of the band is immediately indicated in all of these situations. Deflation of the band may alleviate excessively rapid weight loss and nausea and vomiting. Reoperation to reposition or remove the device may be required.
Rapid weight loss may result in symptoms of malnutrition, anemia and related complications (i.e., polyneuropathies). Deflation of the band may alleviate excessively rapid weight loss.
Rapid weight loss may result in development of cholelithiasis which may require cholecystectomy.